Message Board



One-stop data solution to help all roles efficiently use clinical research data.
Slow validation system launch speed, squeezing the validation window and leaving data quality and clinical safety risks insufficiently understood.
Weak system extensibility, unable to jointly validate problematic data using multiple validation methods.
Weak risk visualization capability and inadequate ability to provide security event risk warnings.
Rich built-in risk validation models for a fast validation experience
Integration and management of medical risks, providing professional clinical data analysis and visualization trend insights. This further reduces events risks of research safety and ensures data consistency and accuracy.
Combining adaptable external data integration solutions, we offer a one-stop clinical data governance solution for clinical data and risk monitoring.
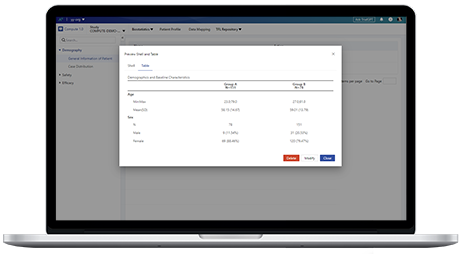
Customer D, a leading medical device company in China, faces the following challenges in clinical data management:
Inefficient communication and integration due to offline circulation of trial design specifications among departments.
System interface failures hinder validation efficiency and prevent tracing of problematic data.
Lack of sedimented validation experience hampers reuse and initialization across projects.
AuroraPrime's clinical research data statistics and analysis solution addresses the customer's actual needs from a systemic level:
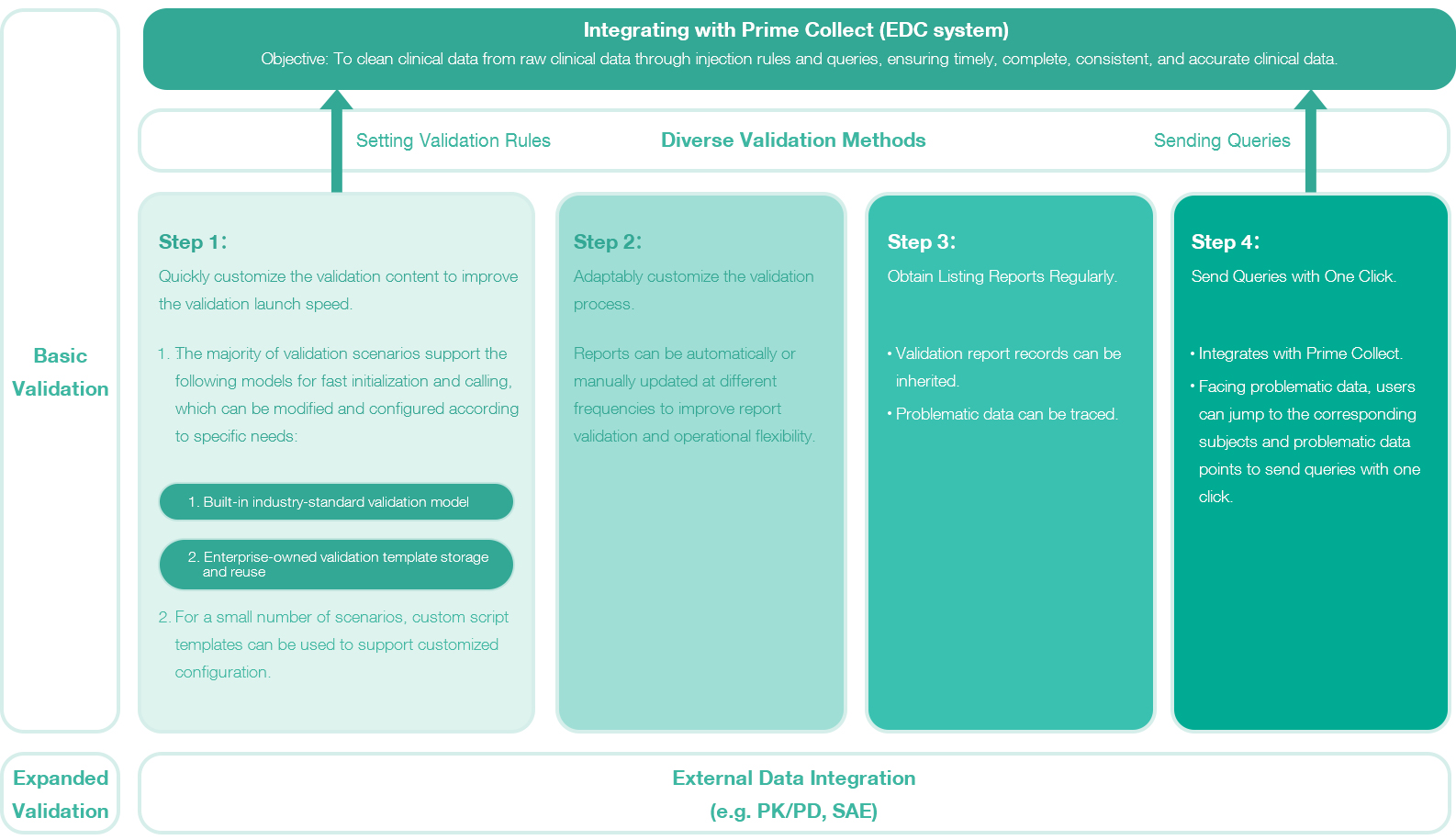
Built-in industry-standard data validation templates greatly simplify data validation configuration and reduce code configuration, improving validation report launch speed. Additionally, validation models can be reused among clinical projects, significantly shortening clinical data governance cycles.
VSupports customizable validation frequency and report result generation. The data management department can automatically or manually update reports at different frequencies according to different needs, improving report validation and operational adaptability. Validation report records can be inherited, and problematic data can be traced.
Integrates with Prime Collect (the EDC system of AuroraPrime) to send queries with one click. Facing problematic data, users can jump to the corresponding subjects and problematic data points to send queries.
Supports external data integration to expand system data validation capabilities. For example, external data such as PD/PK, SAE, IRC, and laboratory data can be jointly validated with the system, further expanding the system's data validation capabilities and scope.

Prime Compute is a one-stop data governance solution for clinical data and risk monitoring, leveraging rich built-in risk evaluation models and high scalability. It helps us manage medical risks and provides professional clinical data analysis and trend visualization. With Prime Compute, we effectively reduce the risk of safety incidents in research, ensuring data consistency and accuracy, and improving operational efficiency of trials

Improve the efficiency of clinical research and development and
bring innovative products to market faster.
