Message Board



Our comprehensive solution facilitates seamless connectivity and rapid execution of clinical trials, starting from the design phase with the "Protocol" and extending to the online data collection system "EDC." With structured protocols, standardized data, and automated database creation, our one-stop solution empowers efficient end-to-end clinical research processes.
Long overall time span and high costs
Possibility of protocol amendment during EDC database construction and deployment, leading to information asymmetry risks
Differentiation in the mapping of protocols and EDC databases, making it difficult to structure it as enterprise-level data
Significant reduction in the time from protocol writing to EDC database deployment
One-stop completion of protocol to EDC database deployment, with data exchange between upstream and downstream, and timely intelligent prompts for content and information updates
AuroraPrime's Clinical Logic Engine based on AI technology can help enterprises to map logic and create a dedicated "Library" for the process from protocol to EDC database deployment.
Customer B, a leading innovative pharmaceutical company in China, has identified and aims to address the following challenges:
Time-consuming mock CRF design involving multiple parties, including the CRO data management team, leading to delays in the ethical submission process after finalizing the protocol.
Low collaboration efficiency between the client and external teams (CRO), impacting the process from protocol finalization to CRF finalization, database construction, and UAT testing, often necessitating rework.
Uncontrollable delays and issues arising from changes in customer project milestones, resulting in insufficient time for external teams (CRO) to process tasks such as database construction or UAT testing.
AuroraPrime's rapid deployment solution for clinical trials, designed to address the client's actual problems, meets the client's needs at the system level with the following functions:
AuroraPrime's EDC database construction module (Prime Construct) supports the automated generation of Mock CRF by inheriting the protocol content from Prime Create (which also supports direct import of external protocol documents), solving the long cycle problem from protocol finalization to Mock CRF completion.
When the protocol amendment happens, Prime Create tags the "changed content" and passes it to the AuroraPrime's EDC database construction module (Prime Construct), providing intelligent content difference prompts and helping the CRF finalization work of the DBD team to be more efficient.
Prime Construct module revolutionizes database construction by introducing a new declarative rule system that replaces complex backend programming languages. Using a user-friendly click-and-drag interface, users can efficiently and conveniently complete database construction tasks. Additionally, automated functions like DVP generation, edit checks, and test case execution significantly reduce EDC deployment time.
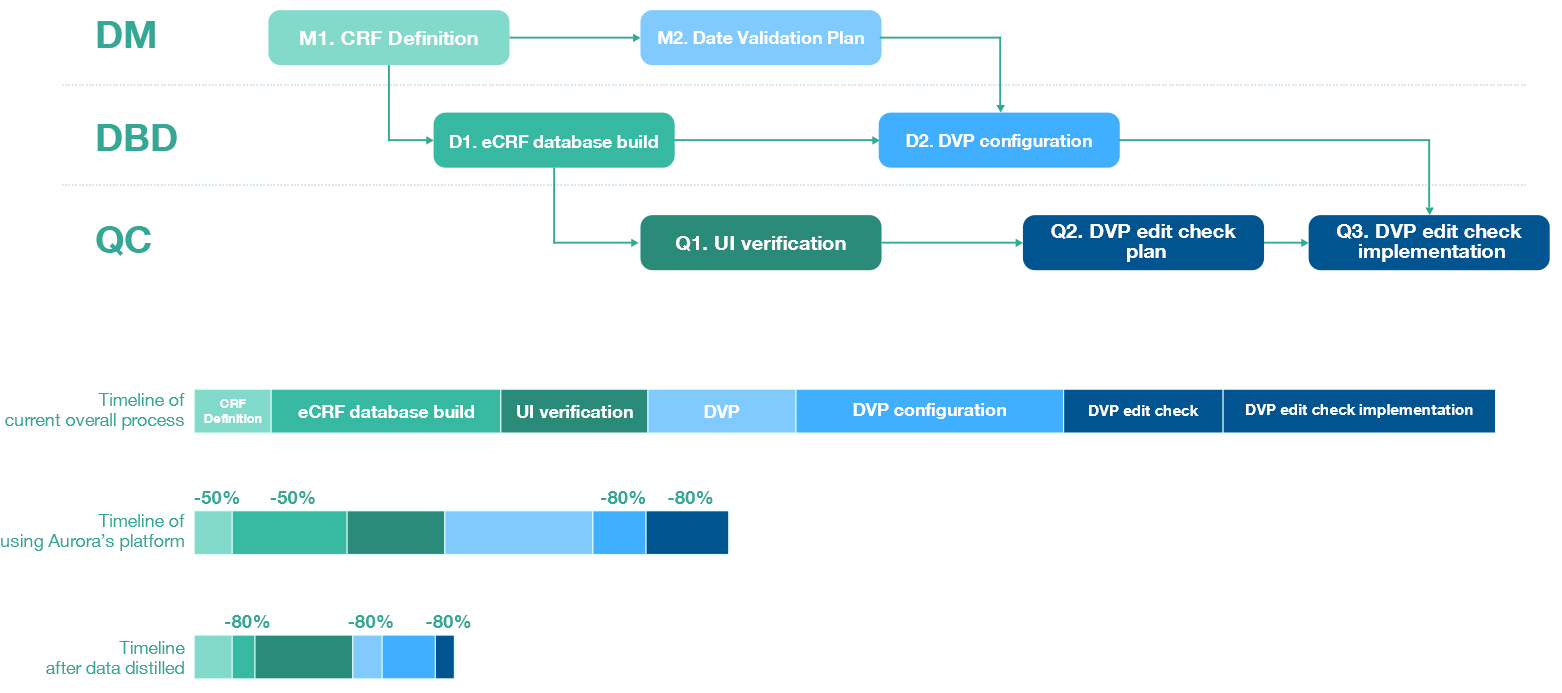
Thanks to AuroraPrime's solution, Customer B's EDC deployment time has been significantly optimized, reduced to less than 50% of the original duration. The average error rate for inter-departmental collaboration has also decreased to below 25%, providing sufficient time for quality assurance and user experience before clinical data collection. Customer B has solved the previous challenges from protocol to EDC deployment process in a one-stop manner.

By integrating protocol drafting with automated database creation and EDC deployment, AuroraPrime's solution achieves end-to-end collaboration between medical and data teams, ensuring efficiency and continuity throughout the entire process. System integration reduces the communication cost of cross-departmental collaboration and enables standardized and structured data and content to flow efficiently across departments. Our expert team can focus their core working time on creating innovative and valuable content and ensuring quality, thanks to the seamless and efficient workflow provided by AuroraPrime.

Improve the efficiency of clinical research and development and
bring innovative products to market faster.
